

Solid
Solid

Solid:
A phase of matter where the atoms are arranged in a stable three-dimensional structure that resists
forces. The Atoms can be arranged in three main ways: crystalline, amorphous, and quasicrystal.
Types of Solids
Crystalline

Crystalline solids are substances whose atoms align in an orderly and periodic manner. The atoms connect in
all known ways: metallic, ionic, covalent, and molecular. An example of a crystalline solid is the calcium
in your bones.
Metallic
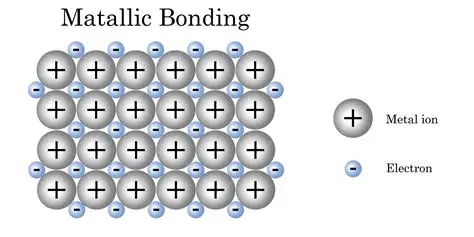
A metallic solid is the solid form of one or more metallic elements (of which make up around a third of the
periodic table); almost all metals are conductive of heat and electricity because of free flowing electrons
between the atoms. Metals tend to be reflective and malleable. The calcium in your bones is a type of
metalic solid.
Ionic
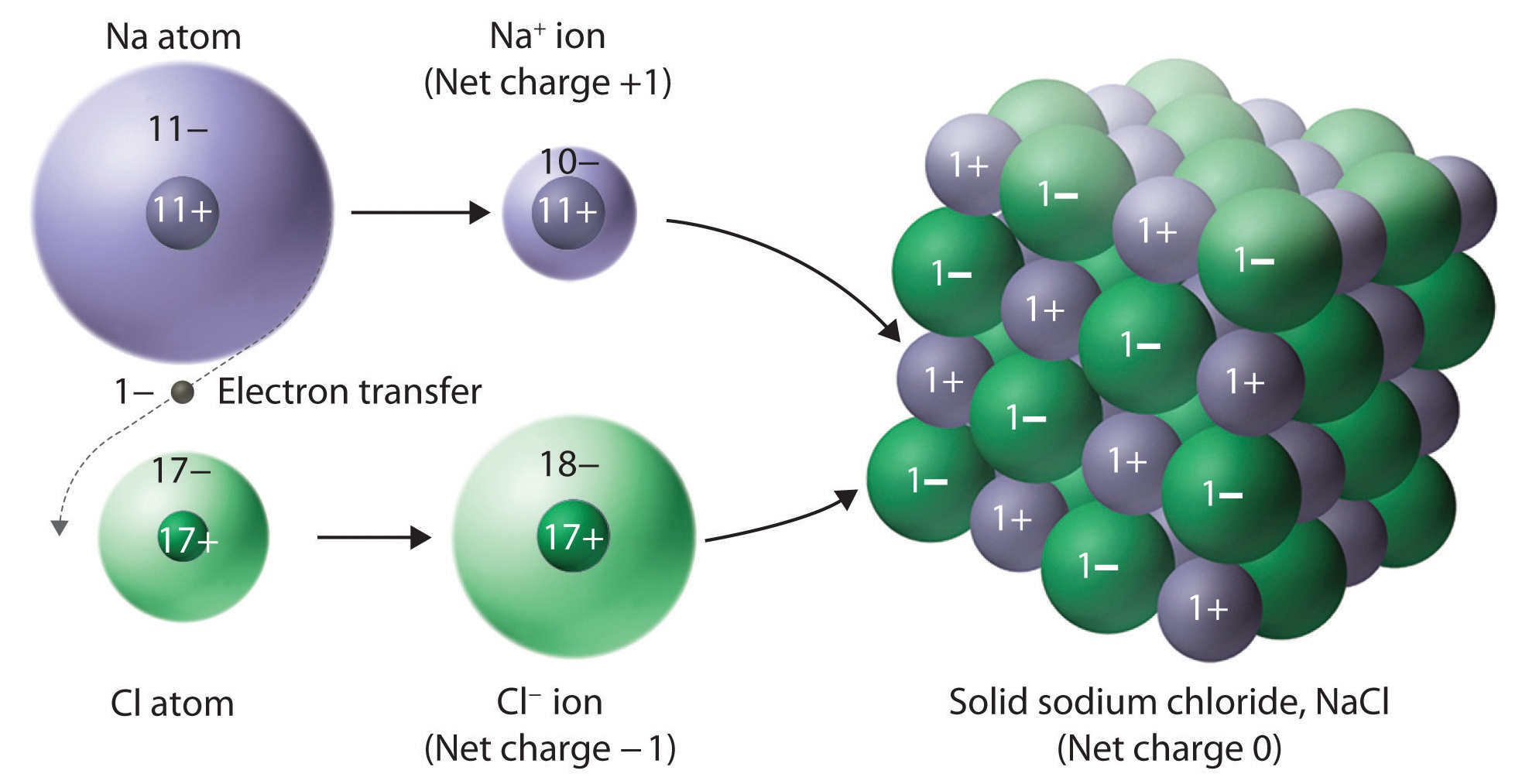
Ionic bonds are when an atom with an extra valence electron meets another element missing one electron, and
instead of sharing the electron, they exchange it, causing them to have opposite charges and bonding them
together. Solids made of ionically bonded atoms arrange themselves so their poles are flipped every pair.
The positive ion (the one that gave up its valence electron) and the negative ion (the one
that took it) cancel each other's charges out and make each other relatively inert. They become inert
because both their outer shell of valence electrons are full. Salt is a type of Ionic solid.
Covalent
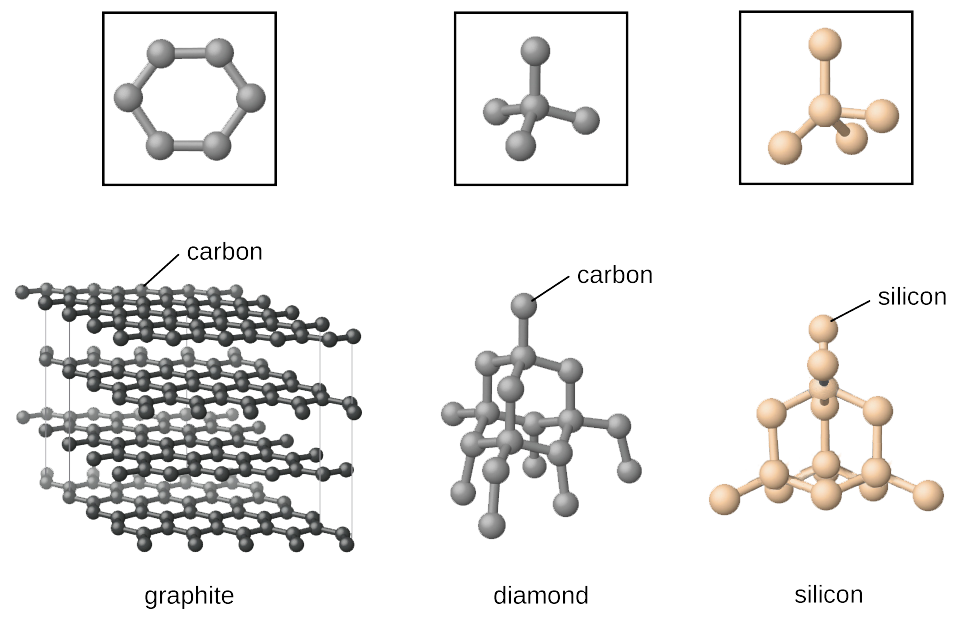
Covalent bonds occur when two atoms share an electron with each other. These bonded atoms can form solids,
which tend to be hard and brittle. A great example of a covalent solid is graphite.
Molecular
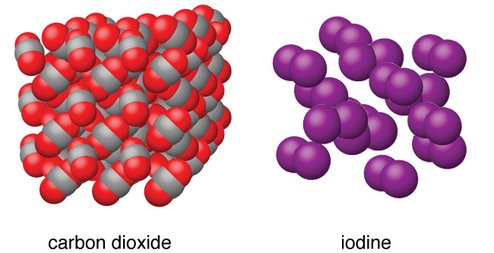
Molecular solids are solids with intermolecular binding; the binding tends to be weak. An example is frozen
argon.
Amorphous

Amorphous solids don't have a consistent crystalline structure. Amorphous solids consist of the same four
types of binding as crystalline solids, but in an irregular form. Some examples of amorphous solids are
glass, hard candy, poly lactic acid, and gel.
Amorphous Vs Crystalline Structure
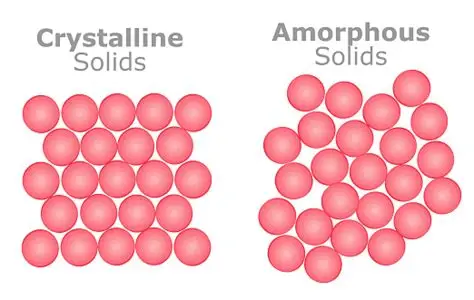
Average Amorphous Structures
Amorphous solids often don't go straight to liquid form when heated, but rather have a longer in-between
state; these properties make amorphous solids like glass and plastic great for making things.

Quasicrystal

Example Quasicrystal

Quasicrystal solids are identifiable by their lack of a uniform structure that still has a sense of
constancy. The bonds of their atoms tend to have some form of radial symmetry, like the example above. The
best way to describe their internal structure is that they are a bunch of three dimensional shapes arranged
in a pattern with no obvious period (for the possible uninformed a period is the start and finish of a
repeating thing, like a sin wave, we here at Phase Phrogs do not know if other people are using this word
like we are right now). Overall, these solids are very interesting, but we have wasted our time and the
website is to be presented to the shareholders tomorrow, and we are nowhere near quota. If you would like to
read more about quasicrystal solids, click
here.
Examples are many different alloys with aluminum mixed in.
Elite Ball Knowledge
Most metals are conductive, but it is not just metals that conduct, other types can to.
Some
solids are conductors at hot temperatures; these solids are called
semiconductors.
I want to eat molten glass. (it looks like candy)
Liquid

When heat is applied to a solid, it turns liquid.